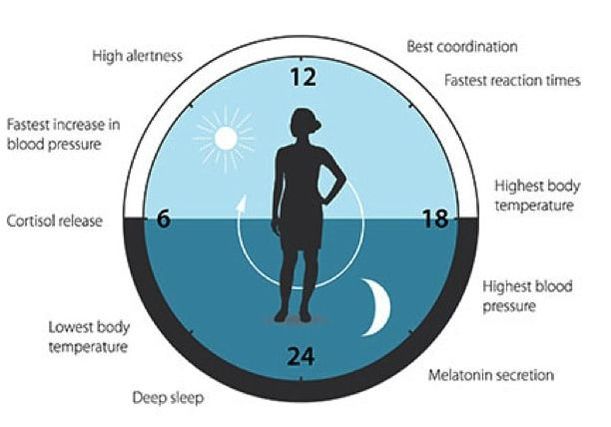
Circadian rhythm or clock refers to a complex mechanism underlying the capacity of living organisms (plants and animals including us) to adjust several of our body processes to the sunlight during the day and darkness at night in 24h (solar) oscillating cycles. This figure taken from the press release of the 2017 Nobel prize awarded this past October 2nd shows how the circadian clock helps us adapt our physiology to the different phases of the day and regulates sleep patterns, feeding behavior, hormone release, blood pressure, and body temperature.
https://www.intechopen.com/books/molecular-mechanisms-of-the-aging-process-and-rejuvenation/circadian-clock-gene-regulation-in-aging-and-drug-discovery
Several genes have been identified that participate in the main feedback loop responsible for circadian cycles. A transcription factor formed by a combo of two proteins (BMAL1 and CLOCK) controls expression of the repressor genes PER and CRY (and also transcription of other clock-controlled genes), which together form a complex with other proteins to repress BMAL1-CLOCK function on chromatin. This is known as negative feedback (=repression or silencing of genes) that inhibits synthesis of the PER and CRY genes. Once PER and CRY protein concentrations are reduced significantly, negative feedback ends and a new molecular cycle starts. This loop in almost all cells/tissues results in a self-sustaining molecular 24 h rhythm, with specific genes and resulting activated/repressed processes involved in different tissues. The complex genetic regulation that underlies this circadian clock working smoothly in different types of cells involves a great deal of epigenetics (another topic awaiting a Nobel Prize- for details on this see my homepage) occurring at the level of histones, DNA and RNA modifications.
Experimentally, to determine whether a cell is cycling (or whether its circadian rhythm is intact) expression of a clock-related gene is measured, for example BMAL1 expression in a young normal mouse heart shown in the figure below.
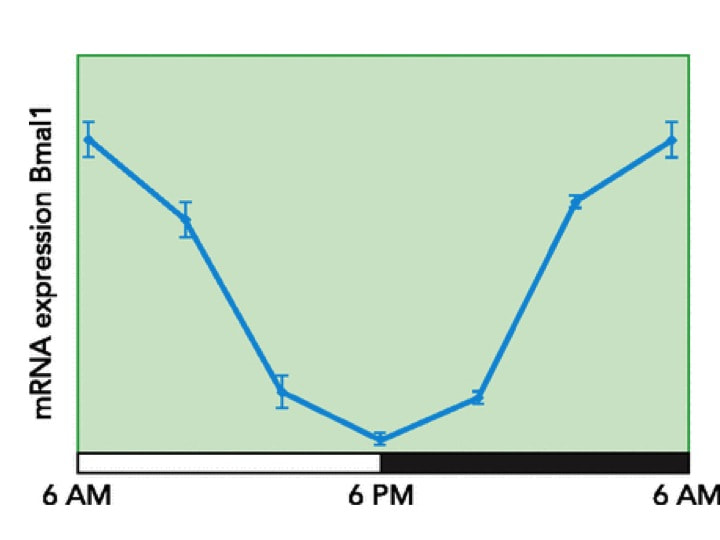
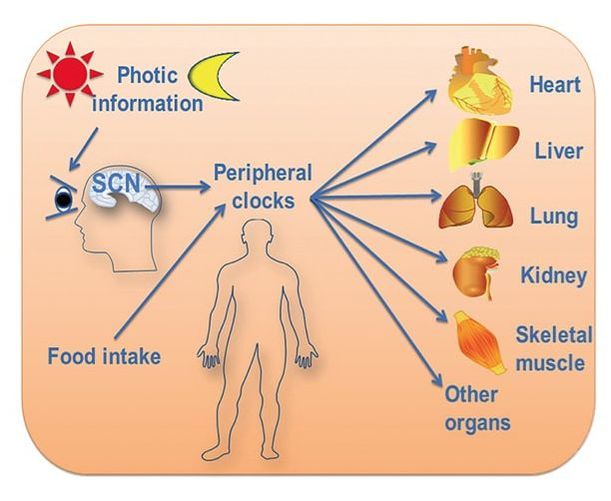
As explained in my home page, thanks to molecular biology and applied fields, once important genes in a biological phenomenon under study are identified, awesome tools are generated (in this case in mice) by mutating or deleting these genes and observing the effect in physiology, behavior, gene expression, cell biology, etc- whatever is now disturbed in these mutants are processes in which the mutated genes (when not mutated) have essential roles. Some “explant” experiments have been used to show that cells taken away from the whole organism can maintain the circadian rhythm autonomously, and this gets affected when clock genes are mutated- these “arrhythmic” effects upon mutations in clock genes have been reported in lung, liver, cornea, kidney, fibroblasts and the SCN.
It is not difficult to infer then how important it is to help our bodies keep this internal clock that provides the rhythm for us to function daily with as little disturbance as possible. But modern lifestyle results in a number of disruptions: jetlag/traveling, night shift jobs, irregular eating and sleeping patterns, light pollution (from the street or our tablets/phones/screens) that affect and confuse our internal cellular clocks as well as the central master SCN regulator. Even before clock genes were considered mechanistically, daily patterns for some diseases symptoms severity were observed: rheumatoid arthritis patients usually feel worse in the morning (thus, nighttime rather than daytime administration of slow release medication is an effective treatment) while osteoarthritis patients feel worse throughout the day. Many studies have demonstrated that shift workers and people with chronic sleep disruption have an increased risk to develop certain conditions such as obesity, type 2 diabetes, hyperlipidemia, high blood pressure, cancer and cardiovascular disease, and they can experience higher levels of inflammation.
Knowing what the rhythm is in special cells/tissues in our bodies can inform medicine approaches, one example of this is the so called chronotherapy, consisting of synchronizing drug administration with circadian rhythms to achieve maximum therapeutic effect and minimum side effects. We already know that many genes that encode important metabolic enzymes including some that process drugs used as medicine show circadian rythms in cells. Medication could then be administered when it is expected to have maximum effect on its target (assuming the drug gets access soon after administration) and/or when enzymes that degrade them in our bodies are at the lowest level.
Another interesting research angle is the circadian rhythm disruption associated with eating schedules (eating/fasting) leading to obesity. The hormone insulin, produced in the pancreas, enhances transfer of glucose from the blood (after eating) into liver, muscle and fat cells, and blocks fat burning. Studies in mice have shown that tissues are somewhat “resistant” to insulin during the inactive/fasting phase of the circadian cycle (during which glucose is converted primarily into fat) and sensitive to insulin during the high activity/eating phase. Mice mutant in clock genes get “locked” into an insulin-resistant mode characteristic of the inactive/fasting phase, and when fed a high-fat diet they gain more weight and fat than normal mice. Interestingly, the same thing happens to normal mice placed under constant light, which may explain the high rate of obesity and diabetes among night-shift workers.


 RSS Feed
RSS Feed