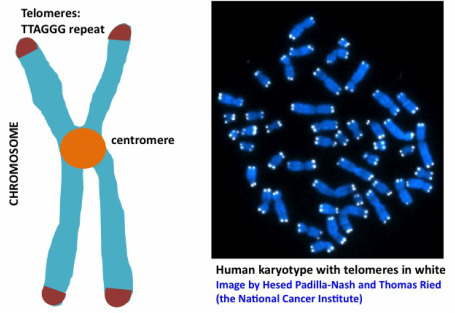
Telomeres refer to the ends of linear chromosomes (from Greek: telos = end, meros = part) consisting of little caps that contain no genes but specific repetitive DNA sequences as shown in the figure below on the left -a hexanucleotide sequence that goes on and on... They can be visualized under the fluorescence microscope by using appropriate staining techniques, shown on the microscopy photo below on the right.
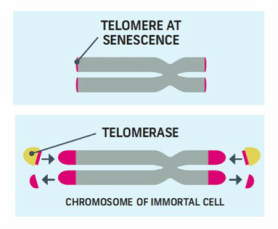
Telomeres have been shown to be very important in maintaining chromosome integrity. The mechanism by which these last bits of DNA on either chromosome end are replicated and maintained at a certain length is extremely complex, requiring a group of specialized RNA and proteins together to form the enzyme telomerase, responsible for lengthening telomeres. Telomerase was discovered and studied in different organisms for the first time in the '90s. The telomerase complex includes a "reverse transcriptase" enzyme (for details on these enzymes check my previous post on PCR) called TERT for TElomere Reverse Transcriptase.
Because "DNA breaks" are very bad for the cell (these can be induced by chemical agents, UV and other types of radiation, etc) and have to be sensed and repaired by specialized cellular processes and enzymes, telomeres (which are essentially pieces of DNA exposed at the tips of chromosomes) have to be protected by what is called "capping" so they are not "repaired" and sometimes fused with other chromosome ends in an attempt to fix what the cell might wrongly see as a DNA break. There are proteins bound to telomeres that block access to them, called "shelterins" during times of the cell cycle in which they might be seen as broken chromosomes.
Because of the way telomere replication occurs, they get shorter and shorter with each cell division, until they reach a critical shortness resulting in chromosome instability, cell senescence and eventually cell death. This mechanism is sometimes referred to as a cellular "clock" that regulates how many times a cell should divide before dying.
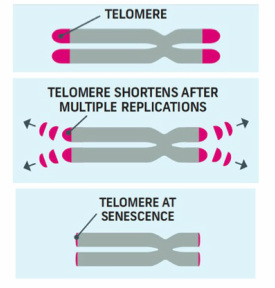
Aging of course is a topic that everyone is interested in, especially when anti-aging possibilities arise. When telomere length and telomerase were studied further, some commercial products appeared in the market such as supplements and anti-aging creams claiming development of new formulas containing agents that could sow down aging based on counteracting the short telomeres effect or providing telomerase, such as a $1,500 skin cream (http://tmagazine.blogs.nytimes.com/2010/04/22/miracle-worker/).
In 2009, the Nobel Prize in Physiology or Medicine went to Elizabeth Blackburn, Carol Greider and Jack Szostak for their studies on telomeres. A short interview with Elizabeth Blackburn on new and exciting applied research on telomeres if found at:
http://www.scientificamerican.com/article.cfm?id=blackburn-elizabeth-telomeres-anecdotes-from-nobel-prize-winner
A malignant, cancer cell which divides in an unregulated manner, usually has levels of telomerase activity higher than equivalent cells form the same tissue which are not malignant (these cells in fact have low or no detectable levels of telomerase unless they are stem or progenitor cells). This has been shown to be the case not for early forms of cancer but more advanced ones; telomerase can be detected in approximately 90% of all malignant tumors which makes it a highly attractive therapeutic target. These cells are thought to activate telomerase to lengthen the otherwise critically short telomeres which would induce senescence and death. The presence of active telomerase confers the immortality intrinsic to cancer cells which can divide uncontrollably.
As a final remark, just to emphasize how much better it seems for us to have longer rather than short telomeres, numerous studies that have looked at people with different conditions have found that, besides aging and cancer, short telomeres seem to be associated with the following:
* earlier death (Denmark)
* short sleep duration
* phobias, anxiety, depression, stress, schizophrenia
* childhood chronic or serious illness (Finland)
* several diseases
However, as with all correlations found in clinical research, we have to be very cautious when interpreting these, as they don't necessarily indicate a CAUSAL relationship in one particular direction. All these conditions might affect the way our cells divide and replicate their DNA, resulting in more DNA damage accumulated with time, and therefore shorter telomeres could be a downstream effect.


 RSS Feed
RSS Feed