After a person is bitten by an infected mosquito, the incubation period ranges from 1-4 weeks. The first symptoms are usually very similar to the flu: aches and pains, fever, headache. After a few days, the typical malaria symptoms occur: chills followed firstly by a high fever for a few hours, and then by profuse sweating. In between these episodes, the patient may feel well. Because the initial symptoms overlap with those of other diseases (both tropical which often include fever, as well as non exclusively tropical like the flu), malaria tends to be overdiagnosed in malaria endemic areas (underdeveloped, tropical countries mostly), and in more developed, temperate areas where it is not prevalent (patients may be travelers returning from regions where malaria is endemic) it could be underdiagnosed as medical doctors are not used to thinking of the possibility of people being exposed to certain tropical diseases.
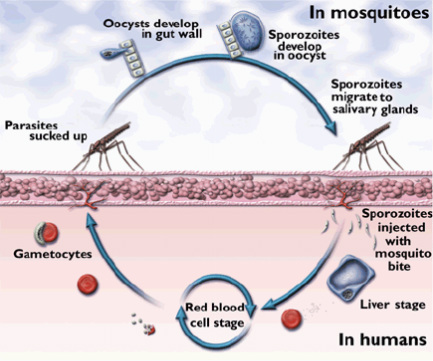
Malaria is a complicated disease at many levels. For example, there are different parasite species that can infect humans. The two main parasites in the genus Plasmodium that cause malaria in humans are P. falciparum and P. vivax. In Africa, the main species is P. falciparum, and in Asia it is P. vivax, athough in Asia patients are found infected with either and also with both (mixed infection) species. The life cycle of Plasmodium comprises different stages in the two animals hosts (human and mosquito- see figure below), and this becomes a major problem when treating the disease, as anti-malarial drugs, when administered to humans, may not kill all parasite stages. P. vivax can further complicate the cycle by producing dormant stages (hypnozoites) in the liver that 'reactivate' several weeks, months or years later.
When trying to eliminate malaria from specific regions, coordinated efforts including vector control have to be deployed to reduce the mosquito population and hence transmission. These mosquito control measures are mainly insecticide-treated mosquito nets and indoor residual spraying, which can be very effective in preventing malaria among children in endemic areas.
Believe it or not, in the early 20th century, before penicillin was discovered and available to cure many diseases including neurosyphilis, doctors would give patients malaria on purpose by injecting blood with parasites from ill patients, which proved to result in remission from neurosyphilis in many cases. After a few fever episodes, the patient will be given quinine, the effective antimalarial drug at the time. Empirical observations had revealed that some of these (by then considered terminal) patients would get better and even go into remission after experiencing infections that elicited high fevers in the patient. Neurosyphilis is an advanced syphilis stage which attacks the nervous system, causing paralysis and psychosis, and which may have been an important factor in the rising number of patients in asylums in the earlier 20th century. Julius Wagner-Jauregg won the 1927 Nobel prize for his discovery of the therapeutic value of malaria inoculation in the treatment of neurosyphilis.
Antimalarial drug resistance: a never-ending problem?
If there is a disease that has consistently shown through several decades of different treatments that these, especially when given as mono therapy, can lead to development of drug resistance, this is malaria (for details on how drug resistance arises in infectious organisms, see my blog post “drug-resistant infections”). Different drugs have been successfully used (when initially introduced) to treat and prevent malaria, with each gradually losing its effectiveness with time as drug resistance emerged. Quinine was the first such drug, introduced in the 17th century as an antimalarial (which is debatable as there is some evidence that Peruvian Quechua Indians were aware of its anti-fever properties long before). As a curious note, tonic water contains large amounts of quinine, and this could be the reason behind its popular use in drinks such as gin and tonic in British colonies. Quinine potency began fading in the 1940s, and it was replaced with chloroquine. Once evidence of emergence of drug resistance to chloroquine was revealed in the late 1950s and early 1960s, a new combination treatment, sulfadoxine-pyrimethamine (SP) was introduced and used for two decades until it was partially replaced by mefloquine due to SP resistance reported. Resistance to mefloquine appeared in the 1980s.
Today, largely due to research led by Professor Nicholas (Nick) White, WHO recommends artemisinin-based combination therapy (ACT) as the main malaria treatment option. ACT consists of a combination of drugs containing one artemisinin derivative and a slower-acting partner, such as SP or mefloquine. Artemisinin, also known as Qinghaosu, is isolated from the plant Artemisia annua, sweet wormwood, a herb employed in Chinese traditional medicine. There are different ACTs, the exact composition of each depending on the geographic area and associated drug resistance patterns. The reason for this combo is to prevent drug resistance: the artemisinin derivative acts faster on the parasites, killing most of them while the older, slower drugs remain longer in the patient, killing off remaining parasites. There are however, reports of artemisinin resistance emerging on the Thai-Cambodian border. Fear of artemisinin resistance spreading is currently prompting intense research in this area as well as efforts towards malaria elimination to prevent resistance spreading. Containment of resistance early is crucial, as otherwise it could spread through West Asia towards Africa, and once in Africa it would have devastating effects- about 85% of worldwide malaria cases are due to P. falciparum infection, and the vast majority of these cases occur in Africa.
Primaquine and G6PD deficiency
Yesterday, a review on the safety of yet another antimalarial (an old and very effective one) primaquine, which belongs to the 8-aminoquinolines group of drugs is out as a WHO (World Health Organization) document and is downloadable from this link:
http://www.who.int/malaria/publications/atoz/9789241506977/en/
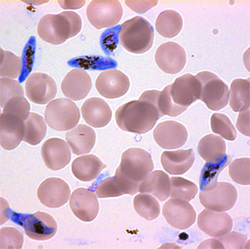
We wrote this review almost 2 years ago to aid evaluate the safety of primaquine when given as a single dose to prevent transmission to the mosquito of the malaria-causing parasite P. falciparum, which results in falciparum malaria and is the main cause of malaria in Africa. Primaquine can be administered in addition to ACT treatment to prevent transmission of the P. falciparum gametocyte stages from the human host to the mosquito vector (P. falciparum gametocyte picture below). This is the current WHO recommendation. However, because there is a risk of hemolytic anemia (when RBCs ‘lyse’ or break up) resulting from primaquine ingestion in individuals who are glucose 6-phosphate dehydrogenase (G6PD) deficient, primaquine use is not as widespread as it could be in malaria endemic areas. There is now evidence that a lower dose of primaquine, which is still effective in killing gametocytes, is safe when given to G6PD deficient individuals, and this is the most recent WHO recommendation.
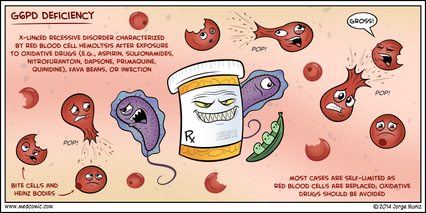

Fava beans. Figure from:
http://www.wisegeek.com/what-is-a-g6pd-deficiency.htm

 RSS Feed
RSS Feed