http://www.nytimes.com/2013/09/17/science/dna-double-take.html?pagewanted=all&_r=0
Chimeric "clones" can arise at different stages of development in either men or women due to mutations in cells which give rise to groups of descendant cells containing these mutations, or as cancer tissues which themselves contain mutated sequences compared to healthy tissues in the same person. There are at least 2 additional ways in which (only) women can think of themselves as chimeras: a combination of different types of cells rather than just the one we started with as a zygote. Although a more precise term would be micro-chimeras, as the group of cells inside some tissues that are different is quite small compared to the rest. One of these two women-specific chimera processes happens to all women, and the second only to those that have been pregnant:
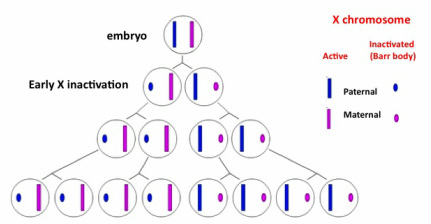
1) X chromosome inactivation (It may be useful for this section to refer to the karyotype figure on my homepage): In females, one of the two X chromosomes in each cell has to be "inactivated" (and in this process, once again, specifically modified histones and other epigenetic factors are involved :-) so only one of them is "expressed" in each cell. This process is a form of what is known as "dosage compensation" and it happens in many animal species to keep the amount of proteins made from X chromosomes the same in males and females. Genetically, one X chromosome in each female embryo came from the mother, and the other one from the father, so X inactivation results in either of these being inactivated in different cells. In males, the sex chromosome pair consists of one X (coming from the mother) and one Y (coming from the father), thus there is no need for X inactivation. X inactivation in females occurs early in development and in a random fashion. The inactive X chromosome suffers "condensation" and is visible under the microscope as a very dense smaller mass called a "Barr body". To put it simply, this condensation into a Barr body makes the genes in the inactive X chromosome inaccessible to proteins that would otherwise be responsible for these genes' expression into active proteins. Once a particular X chromosome is inactivated, the same one will be inactivated in all its daughter cells. However, at the initial stages one cell and its neighbor may have opposite X chromosomes inactivated, resulting in a "mosaic".


 RSS Feed
RSS Feed